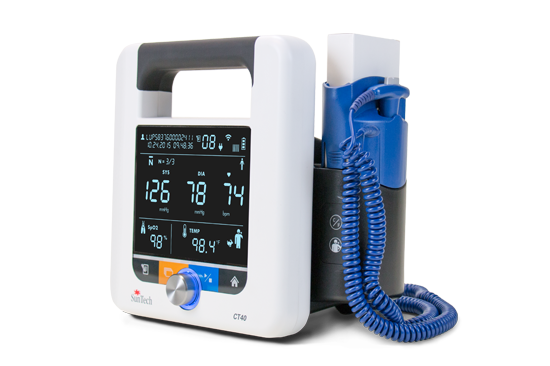
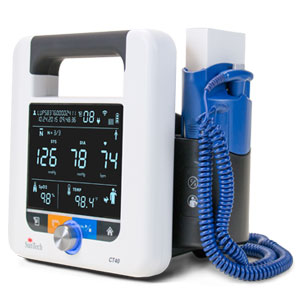
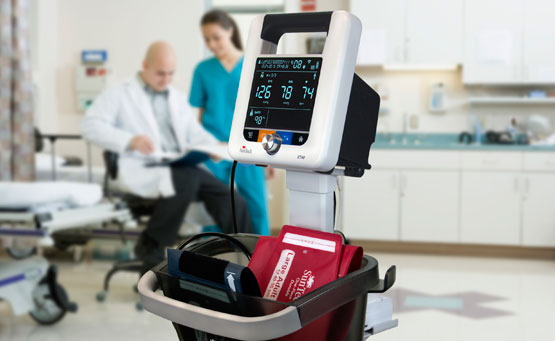
Driven by SunTech’s market-leading Advantage™ BP technology, the new SunTech CT40 is the ideal, affordable solution for clinical-grade spot-check measurements of blood pressure plus vital signs in hospitals and clinics. Ambulatory care, long-term care and low-acuity hospital departments can easily implement this versatile, user-friendly vital signs device that delivers SunTech clinical-grade performance, advanced features and digital connectivity.
Unique to the SunTech CT40, its modular design allows clinicians to easily make adaptations to the device while in the field – adding thermometry, SpO2 and Wi-Fi based solely on the needs of the patient. Other advanced features include the ability to transmit measurement data directly from the device in accordance with HL7 messaging protocols as well as BP Averaging Mode, a key component to accurate blood pressure measurement and used in the recent SPRINT trial conducted by the National Institute of Health (NIH).
Features
Features
With the Suntech CT40, you can count on us to deliver:
Quality: Clinical-grade Advantage™ BP technology
Reliability: Manual SphygMode™ for real-time verification of BP readings without removing the cuff plus built-in memory for up to 99 charting sessions
Accuracy: Customizable BP averaging protocol to ensure accurate, consistent readings (SPRINT recommended)
Connectivity: HL7 messaging protocols for data transfer with network connection through built-in LAN/Ethernet, 2 USB ports or optional Wi-Fi
Flexibility: Modular design for addition of optional temperature and SpO2 modules
Simplicity: Intuitive user interface with LCD display
Options & Accessories
- Covidien Filac® 3000 Thermometry
- Masimo SET® SpO2 (preferred)
- ChipOx Nellcor®-compatible SpO2
- Mobile Stand
- Barcode Scanner
- Wi-Fi

Need more information or pricing? Request a quote here and learn how we can be the Difference in Clinical Grade for you!
Documents
Documents
Downloads:
IHE Integration Statement English 11.1 KB
IHE Integration Statement for the SunTech CT40
- Part Number
- 1
- Revision
- 1
- Document Type
- 1
- Changed
Quick Reference Cards English 298.05 KB
SunTech CT40 Quick Reference Cards
- Part Number
- 82-0145-00
- Revision
- E
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:27
SunTech CT40 Advanced Configuration Application Portuguese 1.58 MB
Advanced Configuration Application for the SunTech CT40
- Part Number
- 1
- Revision
- 1
- Document Type
- 1
- Changed
- Wednesday, 07 September 2016 11:46
SunTech CT40 Advanced Configuration Application Spanish 1.61 MB
Advanced Configuration Application for the SunTech CT40
- Part Number
- 80-0072-04
- Revision
- A
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:39
SunTech CT40 Advanced Configuration Application Italian 1.6 MB
Advanced Configuration Application for the SunTech CT40
- Part Number
- 80-0072-03
- Revision
- A
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:40
SunTech CT40 Advanced Configuration Application German 1.62 MB
Advanced Configuration Application for the SunTech CT40
- Part Number
- 80-0072-02
- Revision
- A
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:41
SunTech CT40 Advanced Configuration Application French 1.6 MB
Advanced Configuration Application for the SunTech CT40
- Part Number
- 80-0072-01
- Revision
- A
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:41
SunTech CT40 Advanced Configuration Application English 1.97 MB
Advanced Configuration Application for the SunTech CT40
- Part Number
- 80-0072-00
- Revision
- C
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:56
SunTech CT40 Brochure English 750.8 KB
SunTech CT40 Brochure
- Part Number
- 82-0141-00
- Revision
- D
- Document Type
- MA
- Changed
- Monday, 01 May 2023 12:38
SunTech CT40 Brochure Portuguese 987.16 KB
Potuguese Translation of the SunTech CT40 Brochure
- Part Number
- 82-0141-05
- Revision
- C
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:53
SunTech CT40 Brochure Spanish 971.95 KB
Spanish Translation of the SunTech CT40 Brochure
- Part Number
- 82-0141-04
- Revision
- C
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:53
SunTech CT40 Brochure Italian 961.57 KB
Italian Translation of the SunTech CT40 Brochure
- Part Number
- 82-0141-03
- Revision
- C
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:54
SunTech CT40 Brochure German 965.87 KB
German Translation of the SunTech CT40 Brochure
- Part Number
- 82-0141-02
- Revision
- C
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:55
SunTech CT40 Brochure French 980.83 KB
French Translation of the SunTech CT40 Brochure
- Part Number
- 82-0141-01
- Revision
- C
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:55
SunTech CT40 Driver for Windows - 32 bit English 329.67 KB
The 32 bit Windows Driver for use with the SunTech CT40
- Part Number
- 1
- Revision
- 1
- Document Type
- 1
- Changed
SunTech CT40 Driver for Windows - 64 bit English 344.26 KB
The 64 bit Windows Driver for use with the SunTech CT40
- Part Number
- 1
- Revision
- 1
- Document Type
- 1
- Changed
SunTech CT40 Quick Start Guide - Main Unit English 5.27 MB
Main Unit Quick Start Guide for the SunTech CT40
- Part Number
- 82-0143-00
- Revision
- F
- Document Type
- MA
- Changed
- Monday, 01 May 2023 12:01
SunTech CT40 Quick Start Guide - Modules English 5.66 MB
Module Quick Start Guide for the SunTech CT40.
- Part Number
- 82-0144-00
- Revision
- E
- Document Type
- MA
- Changed
- Monday, 01 May 2023 12:00
SunTech CT40 Quick Start Guide - Wall Mount 5.37 MB
CT40 Wall Mount Instructions - Quick Start Guide
- Part Number
- 82-0408-00
- Revision
- C
- Document Type
- MA
- Changed
- Friday, 28 April 2023 12:22
SunTech CT40 Technical Specifications English 259.46 KB
SunTech CT40 Technical Data Sheet and list of Parts and Accessories
- Part Number
- 82-0142-00
- Revision
- C
- Document Type
- MA
- Changed
- Monday, 01 May 2023 12:37
SunTech CT40 Technical Specifications French 270.18 KB
French Translation of the SunTech CT40 Technical Data Sheet and list of Parts and Accessories
- Part Number
- 82-0142-01
- Revision
- B
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:52
SunTech CT40 Technical Specifications German 272.6 KB
German Translation of the SunTech CT40 Technical Data Sheet and list of Parts and Accessories
- Part Number
- 82-0142-02
- Revision
- B
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:51
SunTech CT40 Technical Specifications Italian 266.36 KB
Italian Translation of the SunTech CT40 Technical Data Sheet and list of Parts and Accessories
- Part Number
- 82-0142-03
- Revision
- B
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:51
SunTech CT40 Technical Specifications Spanish 266.65 KB
Spanish Translation of the SunTech CT40 Technical Data Sheet and list of Parts and Accessories
- Part Number
- 82-0142-04
- Revision
- B
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:50
SunTech CT40 Technical Specifications Portuguese 263.51 KB
Portuguese Translation of the SunTech CT40 Technical Data Sheet and list of Parts and Accessories
- Part Number
- 82-0142-05
- Revision
- B
- Document Type
- MA
- Changed
- Monday, 01 May 2023 11:50
SunTech CT40 User Manual French 1.47 MB
French Translation of the SunTech CT40 User Manual
- Part Number
- 80-0067-01
- Revision
- E
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:59
SunTech CT40 User Manual English 1.57 MB
SunTech CT40 User Manual
- Part Number
- 80-0067-00
- Revision
- Q
- Document Type
- MO
- Changed
- Tuesday, 14 May 2024 15:14
SunTech CT40 User Manual Dutch 1.54 MB
Dutch Translation of the SunTech CT40 User Manual
- Part Number
- 80-0067-09
- Revision
- E
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:38
SunTech CT40 User Manual Czech 1.47 MB
Czech Translation of the SunTech CT40 User Manual
- Part Number
- 80-0067-08
- Revision
- F
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:38
SunTech CT40 User Manual German 1.46 MB
German Translation of the SunTech CT40 User Manual
- Part Number
- 80-0067-02
- Revision
- E
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:58
SunTech CT40 User Manual Italian 1.47 MB
Italian Translation of the SunTech CT40 User Manual
- Part Number
- 80-0067-03
- Revision
- E
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:58
SunTech CT40 User Manual Spanish 1.46 MB
Spanish Translation of the SunTech CT40 User Manual
- Part Number
- 80-0067-04
- Revision
- E
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:57
SunTech CT40 User Manual Portuguese 1.46 MB
Portuguese Translation of the SunTech CT40 User Manual
- Part Number
- 80-0067-05
- Revision
- E
- Document Type
- MO
- Changed
- Monday, 01 May 2023 11:56
Videos
Videos
SunTech CT40: Initial Setup of Monitor
This video shows the various parts and accessories that come standard with your SunTech CT40 and how to perform the initial setup.
SunTech CT40: Initial Setup of Mobile Stand
This video shows the various parts and accessories that come standard with your deluxe mobile stand and how to assemble it for use with the SunTech CT40
SunTech CT40: How to Add or Remove SpO2 Modules
This video will demonstrate how to add or remove the optional SpO2 Modules for the SunTech CT40.
SunTech CT40: How to Add or Remove Temperature Modules
This video will demonstrate how to add or remove the optional Temperature Modules for the SunTech CT40.
SunTech CT40: Screen & Control Panel Review
This video walks through the user interface on the SunTech CT40 and reviews the various functions of the button panel.
SunTech CT40: How to Take an Automated or Manual Blood Pressure Reading
This video shows the proper way to measure blood pressure using the SunTech CT40 for both the manual and automatic mode.
SunTech CT40: How to use BP Averaging Mode
This video shows how to use BP Averaging on the SunTech CT40.
SunTech CT40: How to Measure Pulse Oximetry
This video shows how to accurately measure pulse oximetry using the 2 different SpO2 modules used with the SunTech CT40.
SunTech CT40: How to Measure Temperature
This video shows how to accurately measure temperature using the 2 different temperature modules used with the SunTech CT40.
Parts & Specs
Parts & Specs
Technical Specifications
SpO2 Specifications
| Function | Masimo M-LNCS DCI | ChipOx |
|---|---|---|
| Saturation Normal (no motion) | 70-100%Arms ± 2% | 70-100%Arms ± 2% |
| Saturation Motion | 70-100%Arms ± 3% | N/A |
| Low Perfusion | SpO2 +/-2% | N/A |
| Pulse Heart rate | ± 3% BPM | 20-300 BPM: ±3 BPM |
| Pulse Heart rate w/motion | ± 3% BPM | 20-300 BPM: ±3 BPM |
Temperature Specifications
| Function | Covidien Electronic Thermometer |
|---|---|
| Temperature Accuracy | Quick Mode (Oral): ± 0.3°C |
| Temperature Accuracy | Std. Mode (Aux/Rectal): ± 0.1°C |
| Temperature Accuracy | Direct Mode ± 0.1°C |
| Response Time | 4 sec (quick mode); 10-15 sec. (Axillary/Rectal); 60 sec.(direct mode) |
| Resolution | 0.1°C |
Blood Pressure Specifications
| Specifications | Values |
|---|---|
| Range: |
|
| Accuracy (BP): | Meets or exceeds ANSI/AAMI/ISO 81060-2:2013 standard for non-invasive accuracy (±5mmHg mean error with 8mmHg standard deviation). |
General Specifications
| Specifications | Values |
|---|---|
| Conditions for Use: | Operating: 10°C (50°F) to 40°C (104°F) 15 – 90% RH non-condensing - 700 kPa - 1060 kPa. Operating the monitor in an environment at maximum temperature can produce temperatures exceeding 41°C (41.6°C highest recorded) on a patient-applied part. It is up to the operator to determine if this temperature is too high based upon the condition of the patient and, if so, to ensure the ambient temperature of the environment is 38°C or below. |
| Storage: | -20°C (-4°F) to 55°C (149°F) 15 – 90% RH non-condensing - 500 kPa - 1060 kPa. Performance can be affected if used or stored outside the specified temperature, humidity, or altitude listed in the ranges above. |
| Power: | Internal power supply. Input: 100-240 VAC @ 1.5A max, 50-60 Hz. Output +9VDC @ 5A IEC 320 type input connector. |
| Battery: | Type: 7.2v, 6600mAh Internal Lithium-Ion battery Run Time (full charge): ~20 hrs* / 200 BP cycles Charge Time: ~6 hrs @ 23deg Celsius +/- 2deg *Unit with BP, Temp, SpO2, Wi-Fi, barcode scanner |
| Calibration: | The accuracy of cuff -pressure transducers/indicators should be verified bi-annually. |
| Safety Systems: | Independent hardware over-pressure circuit and redundant software overpressure algorithm to limit cuff pressure to less than 300 mmHg (+20/-10mmHg). Independent hardware timing circuit and redundant software timer algorithm to limit the duration of a blood pressure cycle to less than 180 seconds. |
| Dimensions: | Size (without thermometer option): 8” H x 6.75’’ W x 5.2’’ D or 215mm H x 190mm W x 140mm D |
| Standards: | ANSI/AAMI ES60601-1:2005/(R)2012 and A1:2012, C1:2009/(R)2012 and A2:2010/(R)2012, EN 60601-1:2006/A1:2013, IEC 60601-1-2: 2007 EMC, IEC 80601-2-30: 2013, ISO 80601-2-61: 2011, ISO 15223-1:2012, ISO 10993-1, 2009, ISO 10993-5, 2009, EN ISO 10993-10, 2010, ISO 81060-2:2013, EN 50419: 2006, EN ISO 14971:2009, CSA C22.2 No. 60601-1, EN ISO 81060-1: 2012, IEC 60601-1-6: 2013 |
| Classifications: | Equipment Classification: Class IIa per MDD, Class I (Electrical Shock), Continuous mode of operation, CE |
| Ingress of liquid: | Ingress Protection: IPX1: This device is protected against harmful effects of dripping water per IEC 60529. |
Limited Warranty
SunTech Medical, Inc. provides to the original purchaser the following limited warranty from date of invoice:
| Specifications | Values |
|---|---|
| Main unit, SpO2 modules, temp modules: | 3 years |
| OPD Blood Pressure Cuffs: | 2 years |
| Accessories (SpO2 sensors, temp probes, BP hose, etc): | 90 days |
Masimo® and SET® are registered trademarks of Masimo Corporation. Covidien® and FILAC® are registered trademarks of the Covidien AG Corporation. Nellcor® is a registered trademark of Covidien LP Limited Partnership.
Parts & Accessories
Base Unit Contents
- SunTech CT40 Device with BP
- AC Power Cord
- 10’ BP Patient Hose
- Battery
- Quick Start Guide
- Adult Plus OPD Cuff
- Large Adult Plus OPD Cuff
SunTech® CT40 System & Modules
| Description | Part Numbers |
|---|---|
| SunTech CT40 Base unit with BP (no power cord) | 99-0134-00 |
| SunTech CT40 Base unit with BP with AC Power Cord (Americas) | 99-0134-01 |
| SunTech CT40 Base unit with BP with AC Power Cord (Europe) | 99-0134-02 |
| SunTech CT40 Base unit with BP with AC Power Cord (UK) | 99-0134-03 |
| Chipox Nellcor® Compatible SpO2 Module | 98-0411-00 |
| Masimo SET® SpO2 Module (preferred) | 98-0411-01 |
| Covidien FILAC® 3000 Thermometry Module | 98-0411-02 |
Systems Reference Configuration
| Description | Part Numbers |
|---|---|
| BP Only | 99-0134-0X |
| BP, Masimo® SpO2 | 99-0134-0X + 98-0411-01 |
| BP, Nellcor® comp. SpO2 | 99-0134-0X + 98-0411-00 |
| BP, FILAC® Temp | 99-0134-0X + 98-0411-02 |
| BP, FILAC® Temp, Masimo® SpO2 | 99-0134-0X + 98-0411-02 + 98-0411-01 |
| BP, FILAC® Temp, Nellcor® comp. SpO2 | 98-0134-0X + 98-0411-02 + 98-0411-00 |
SpO2
Masimo SET® SpO2 Module Accessories
| Description | Part Number |
|---|---|
| Masimo® M-LNCS DCI Adult SpO2 Reusable Sensor | 2501* |
| Masimo® M-LNC 10’/3.0m Patient Cable | 2525* |
ChipOx Nellcor Compatible SpO2 Module
| Description | Part Number |
|---|---|
| Reusable Adult SpO2 Finger Sensor, ChipOx | 52-0010-00 |
Temperature
Covidien FILAC 3000 Temperature Module Accessories
| Description | Part Number |
|---|---|
| F3000 Oral/Axillary Temp Probe, 9’ | 52-0009-00 |
| F3000 Oral/Axillary Isolation Chamber, Blue | 45-0006-00 |
| F3000 Rectal Temp Probe, 9’ | 52-0009-01 |
| F3000 Rectal Isolation Chamber, Red | 45-0006-01 |
| F3000 Disposable Temp Probe Covers (Qty 5000) | 98-0130-00 |
| F3000 Calibration Plug | 52-0011-00 |
Accessories
| Description | Part Number |
|---|---|
| Deluxe Mobile Stand | 46-0040-00 |
| SunTech CT40 Barcode Scanner (w/USB) with Scanner Mount | 98-0257-00 |
| SunTech CT40/50 WiFi Dual Band USB Dongle | 45-0005-00 |
| 10’/3.0m Patient BP Hose, Bayonet to Bayonet | 91-0028-16 |
| AC Power Cord (Americas) | 91-0003-00 |
| AC Power Cord (Europe) | 91-0003-05 |
| AC Power Cord (UK) | 91-0003-06 |
| Mini-USB to USB Cable | 91-0100-02 |
| SunTech CT40 Rechargeable Lithium Ion Battery | 17-0022-00 |
| SunTech CT40 1D Barcode Scanner | 98-0257-03 |
| SunTech CT40 2D Barcode Scanner | 98-0257-04 |
Blood Pressure Cuffs
Blood Pressure Cuffs (Box of 5)
| Description | Part Number |
|---|---|
| OPD Child (12-19cm) BP Cuff, Bayonet | 98-0600-41 |
| OPD Small Adult (17-25cm) BP Cuff, Bayonet | 98-0600-43 |
| OPD Adult (23-33cm) BP Cuff, Bayonet | 98-0600-45 |
| OPD Adult Plus (28-40cm) BP Cuff, Bayonet | 98-0600-4A |
| OPD Large Adult (31-40cm) BP Cuff, Bayonet | 98-0600-47 |
| OPD Large Adult Plus (40-55cm) BP Cuff, Bayonet | 98-0600-4C |
FAQs
FAQs
What is the expected battery life for the SunTech CT40?
When batteries are fully charged, the SunTech CT40 is designed to run on internal battery power for 120 measurement cycles (BP, temperature and SpO2 measurements) before requiring recharge. When only the bottom segment of the battery symbol is illuminated, there is between 11 percent and 40 percent battery power remaining.
Will the device automatically power down?
When no measurements have been taken or buttons pressed for one hour, the device will automatically power-down and enter the low-power state and is considered “off.”
What is the significance of different beeps I hear during operation?
One long beep (approximately 3-second beep): Successful end of BP measurement.
Three long beeps: Severe hardware error.
One short beep (1-second beep): Power-up or Power-down complete.
Four short beeps: There has been an error.
If you abort a BP measurement by pressing the Start/Stop button, you will hear one short beep. Then after the pressure has been completely dumped from the cuff, you will hear one long beep.
Can pulse oximetry, temperature measurement, a barcode scanner or printer be added later too?
Yes, you can add vital signs modules and accessories to the SunTech CT40 at any time.
Can EMR connectivity be established at a later date if not enabled as part of initial configuration?
Yes, EMR settings can be accessed via the Advanced Configuration Application at any time.
Why are no MAP measurements showing on the display?
The ability to measure MAP must be turned on using the Advanced Configuration Application. It is turned off by default at the factory. If MAP is enabled, the results will alternate with heart rate results on the display screen. Note that MAP results are only validated for use outside of the United States.
Why is no heart rate data showing on the display?
If the device is configured to use the SpO2 sensor as the pulse rate source, and the sensor is not attached to the patient, there will not be heart rate data on the display. Only dashes will appear, or MAP results if MAP measurement is enabled.
How many measurements can be stored in the SunTech CT40 memory?
The SunTech CT40 can store 99 unique sets of measurements. After the 99th measurement has been saved, the next measurement will have the Memory Location Identifier label “01” and will overwrite the most recent measurement (measurement 99). . After all results are cleared from the memory, the Memory Location Identifier also will return to “01.”
Does the CT40 use encryption?
Yes, the CT40 offers end to end SSL encryption from the CT40 to the EMR system. If SSL is selected, no plain text data is transferred between the CT40 and the EMR system. SSL can be selected using the Advanced Configuration Application (see document 80-0072-00-MO).
When data is transferred by WiFi to a hospital information system is the data encrypted?
WPA(2) WiFi encryption may be used to provide encryption on the wireless link for any data transmitted over the WiFi. WPA(2) encryption only encrypts the wireless portion of the transfer, data on the wired hospital network is unencrypted. To ensure end to end encryption, SSL must be used.
Is the data on the CT40 encrypted?
The data contained within the CT40 is not encrypted, but it is protected via password and/or encrypted communication channels.
end faq