![]()
Blog @ SunTech
Advice from the BP Measurement Experts
Clinician Beware of Smartphone Health Apps
 Do you have a smartphone? Have you downloaded any apps that help you track or measure your own vital signs information—like blood pressure, heart rate, or glucose level? If you’re a health care professional, maybe you’ve downloaded apps that help you measure and stay informed about your patients’ vitals. Either way, mobile healthcare-related apps are creating significant buzz within the mobile computing industry. Figures vary for the size of the home health and telehealth markets, but nobody questions that the number is in the billions of dollars, and such sweet numbers inevitably attract lots of bees.
Do you have a smartphone? Have you downloaded any apps that help you track or measure your own vital signs information—like blood pressure, heart rate, or glucose level? If you’re a health care professional, maybe you’ve downloaded apps that help you measure and stay informed about your patients’ vitals. Either way, mobile healthcare-related apps are creating significant buzz within the mobile computing industry. Figures vary for the size of the home health and telehealth markets, but nobody questions that the number is in the billions of dollars, and such sweet numbers inevitably attract lots of bees.
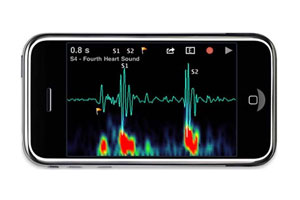
One recent vital signs app that has received lots of attention is the iStethoscope, a mobile app for the iPhone that allows clinicians to use the microphones and processors on their iPhones as virtual stethoscopes. The app has been downloaded millions of times (about 500 downloads per day) and has received lots of press coverage. Another example is a mobile phone developed by a German company called the H’andy Sana 210 that can measure a person’s ECG by just placing two fingers on each side of the phone. The ECG data can then be transmitted to a physician via the cell network. It also allows the user to enter other vital signs data, such as blood pressure, cholesterol, and glucose data.
Smartphones and health apps have definitely added some technological bling to the somewhat staid medical device industry. But in the United States, many of those young, energetic app developers are forgetting about three very important letters: F-D-A. The FDA’s Center for Devices and Radiological Health (CDRH) is responsible for ensuring the safety and effectiveness of medical devices marketed and sold in the U.S., and has established guidelines and requirements for many different kinds of medical products. The burning question, though, is ‘What is a medical device?’
In the case of the iStethoscope, it’s not so clear cut. An iPhone by itself isn’t a medical device. And the iStethoscope app by itself isn’t technically a device either. But when combined, you get “an instrument intended for use in the diagnosis of disease or other conditions, or in the cure, mitigation, treatment, or prevention of disease, in man or other animals.” This is one of the FDA’s definitions of a medical device subject to FDA oversight. Who then must meet the FDA’s requirements? Apple? Peter Bentley, the creator and developer of iStethoscope? Both?
If you are a clinician, these are important questions to ask when considering the use of smartphone health apps. For example, does the app simply track blood pressure data that is entered, or does it turn the smartphone into a blood pressure measurement device? As cool or convenient as they might be, medical devices are regulated for a reason, and you obviously want to ensure that the tools you use are effective and safe for your patients. Until the FDA catches up to such technological trends, the best rule might be, ‘clinician beware.’
Interested in getting more SunTech news, product info, as well as
tips, tricks, and insights from BP experts?
Sign up to get fresh content delivered direct to your inbox.