![]()
Blog @ SunTech
Advice from the BP Measurement Experts
Preventing Medical Mishaps: Understanding the Importance of ISO 80369-5
Medical device manufacturers are subject to many rigorous safety standards to ensure that patient’s lives are not endangered due to equipment related errors. One of these standards, ISO 80369-5, establishes a general framework and guidance for small-bore connectors intended for use in healthcare applications, promoting patient safety by reducing the risk of misconnections between different delivery systems.
ISO 80369-5 is a section of the ISO 80369 series of international standards titled “Small-bore connectors for liquids and gases in healthcare applications” which specifically applies to limb cuff applications. “Small-bore” refers to the size of the opening of the connector, in this case less than 8.5 mm. This article delves into the relevance of ISO 80369-5, its impact, and the steps medical device companies are taking to adhere to this standard.
Why It’s Important
Luer or luer-style connectors have become prevalent in small-bore connection systems on medical products for various health applications. The luer connector is simple in design, robust, and easy to use, so it has been designed into many different products that may connect to or from the patient. Unfortunately, because it has been designed into so many different kind of products, that unintentionally introduced patient risk. Unfortunately, because it has been designed into so many different kind of products, that unintentionally introduced patient risk. ISO 80369-5:2016 was introduced to minimize the risk of accidental misconnection, as the US Food & Administration reported several incidents involving misconnections that caused harm to the patient.
Illustrating the Dangers: A Case Study
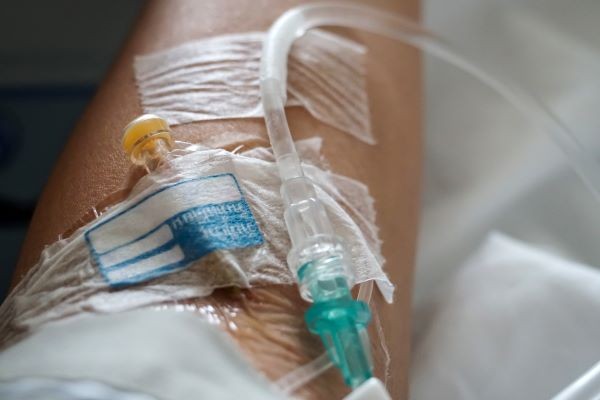
An ER patient had both an IV (no IV fluids had been started yet) and blood pressure cuff attached while undergoing treatment (1). To use the restroom, the patient disconnected all tubing. When she returned, her spouse mistakenly connected the cuff tubing to the IV, delivering approximately 15 mL of air to the IV catheter.
The patient died from a fatal air embolus, despite resuscitation efforts.
Ensuring Compliance: SunTech’s Commitment to ISO 80369-5
SunTech prioritizes patient safety and meeting the highest quality standards. As a leader in blood pressure technology and solutions, SunTech will transition all finished good medical devices to only use connectors that meet ISO 80369-5 (bayonet pictured below).
 |
 |
| Female Bayonet (Rectus) Metal | Male Bayonet (Rectus) Plastic |
FAQs: Your Questions About ISO 80369-5 Answered
Q: What regions will this new connector standard impact?
A: ISO 80369-5 was incorporated into MDR requirements, so it currently affects Europe and anywhere the CE mark is accepted. It is anticipated to be adopted by other regulatory bodies, if not already.
Q: Am I required to change the cuffs I already have?
A: No, however, we strongly encourage your facility to make this change to ensure that you are in compliance with this new standard.
Q: How do I know if my cuff is affected?
A: If your SunTech cuff has the new connectors pictured above, then your cuffs comply with the standard. If the connector you use is not pictured, your cuff is impacted by the standard.
Q: How can I convert my connectors to meet the new standard?
A: Although it is not required to convert cuffs already on the market, existing cuffs can be converted by purchasing an adaptor or hose that has the appropriate connectors. Otherwise, you will need to purchase replacement cuffs with the new connectors.
Q: Will I be able to order more cuffs with the non-compliant connector types?
A: Yes, for a limited time. We are actively evaluating inventory and anticipating a last time buy date of October 31st, 2023.
Please note, information in this article applies to customers who purchase directly from SunTech only. Need to share this information with your team or customer? Download our white paper on ISO 80369-5Download our white paper on ISO 80369-5.
(1) FDA. U.S. FOOD & DRUG. Example of Medical Device Misconnections.
https://www.fda.gov/medical-devices/medical-device-connectors/examples-medical-device-misconnections
Interested in getting more SunTech news, product info, as well as
tips, tricks, and insights from BP experts?
Sign up to get fresh content delivered direct to your inbox.